Medical company guilty of distributing misbranded cancer drug for 13 years



AmerisourceBergen Specialty Group admitted to a court that its subsidiary company, Medical Initiatives Inc (MII), illegally distributed misbranded a cancer drug for 13 years.
They were reportedly shipping the drugs from a facility that wasn’t registered with regulators, and following the conviction, the company has been ordered to pay almost £200m in fines.
The guilty company was found to have been working outside of regulatory review and scrutiny as its facility was never registered with regulators as either as a manufacturer or a re-packager.
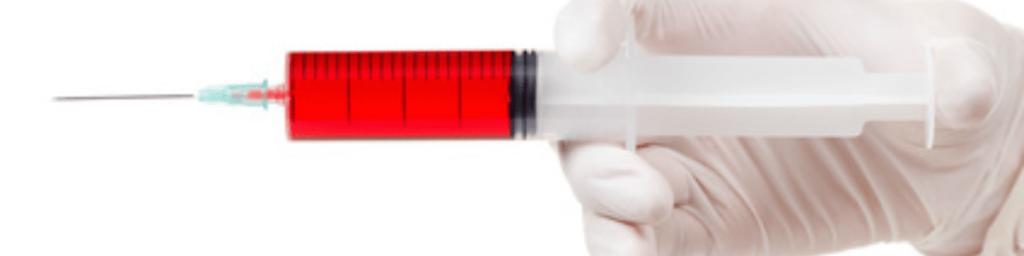
Prosecutors said that the pre-filled syringes were made in an environment that was unclean and unsterile.
Essentially, the company ‘pooled’ the drugs by opening the single-use glass vials and repackaging them into plastic syringes so they could also sell off any ‘overfill’ or excess. In this pooling process, the glass vials were reportedly breached multiple times, increasing the risk of contamination each time when they should only ever be opened once: during administration.
The syringes contained:
The pre-filled syringes containing these cancer-treatment drugs were reportedly shipped to multiple oncology centres, medical practices and physicians. For 13 years, the drugs were used to treat immunocompromised cancer patients including members of the Armed Forces and their families.
According to the Department of Justice, some of the repackaged syringes contained, “particles of foreign matter”.
An agent said:
“Injectable drugs prescribed for patients – especially vulnerable cancer patients – must be pure, sterile and produced in an FDA-compliant facility.”
AmerisourceBergen pointed out that no complaints or injuries were ever filed against the company for the drugs they supplied.
However, there could have been a lot of injuries and potentially even fatalities related to the misbranded and possibly contaminated drugs with no one knowing the cause. As the drugs were used to treat cancer patients who are already very sick and vulnerable, it would be difficult to distinguish any harm arising from the syringes supplied.

Really need to know if you have either a medical, clinical or dental negligence claim to make?
Fill out our super-fast form below and our team will contact you and let you know if you have a case to make a claim for compensation.
Claim NowThe content of this post/page was considered accurate at the time of the original posting and/or at the time of any posted revision. The content of this page may, therefore, be out of date. The information contained within this page does not constitute legal advice. Any reliance you place on the information contained within this page is done so at your own risk.